Sansure Biotech Inc. released its 2021 annual report and 2022 Q1 report. Under the severe challenge of the COVID-19 pandemic, the company independently developed various products and achieved brilliant achievements. This report summarizes Sansure’s 2021 achievements, including revenue, R&D priorities, and significant future direction. Sansure maintains a leading position in the medical industry, with stable revenue and strong resilience. In […]
On May 25, the Monkeypox virus Nucleic Acid Diagnostic Kit developed by Sansure Biotech successfully obtained the CE certification, which means that the kit can be distributed in EU countries and countries that recognize the CE certification. Currently, unusual cases of monkeypox have been reported in several countries, including the United Kingdom, Spain, and Portugal, posing a challenge to human […]
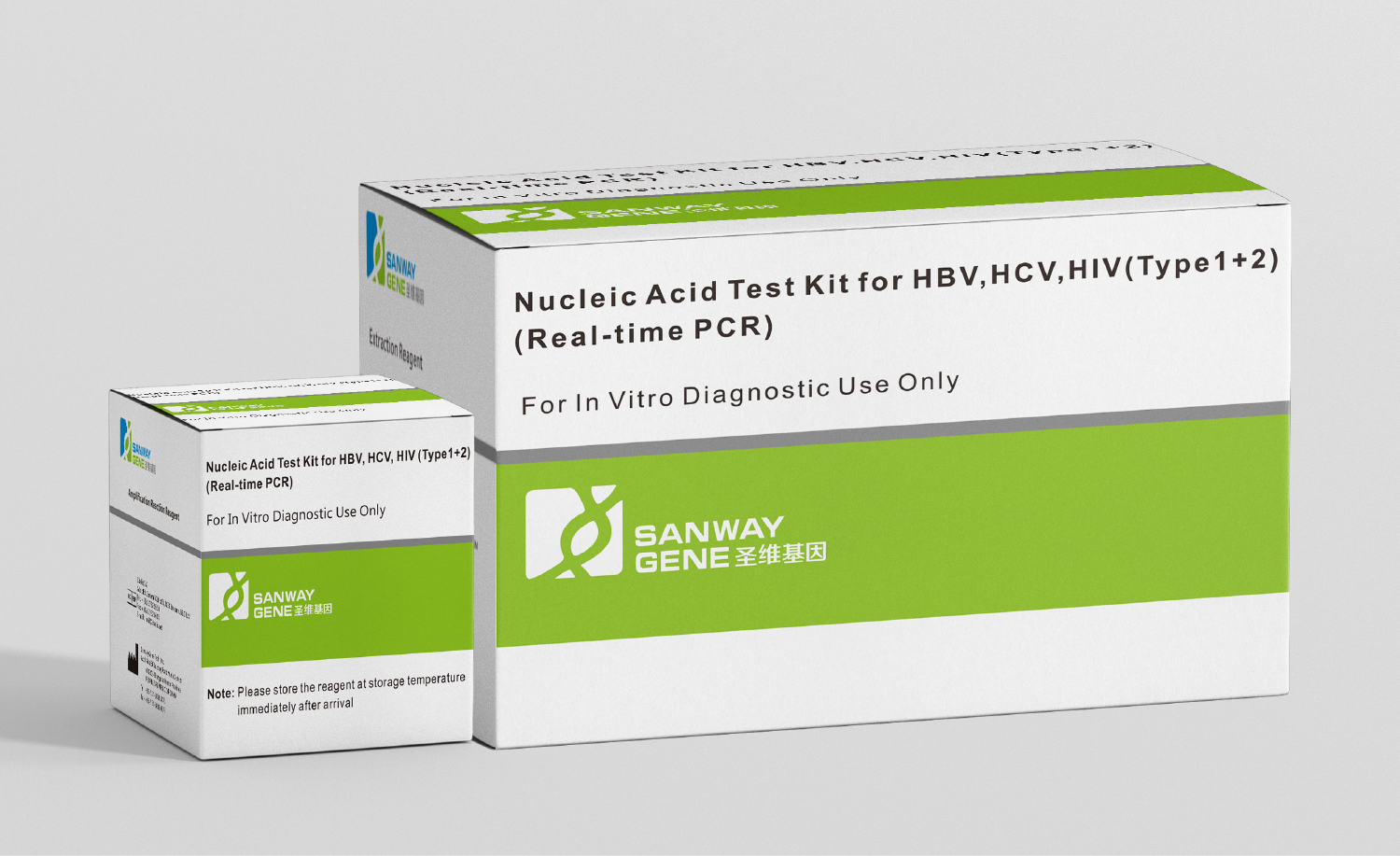
On May 23, the Nucleic Acid Test Kit for HBV, HCV, HIV(Type1+2) developed by Sanway Gene, a wholly owned subsidiary of Sansure Biotech, was successfully certified by the European Union Notified Body (CE List A). It is the fourth CE List A certification obtained by Sansure Biotech following the three molecular kit products of HBV DNA, HCV RNA, and HIV-1 RNA. […]
On May 20, HBV DNA Diagnostic Kit, HCV RNA Diagnostic Kit, and HIV-1 RNA Diagnostic Kit developed by Sansure Biotech Inc. have successfully obtained the European Union Notified Body Certificate (CE-IVD List A), making Sansure the first in China to receive the highest CE certificate for molecular testing reagents. HBV CE certificate (List A) HCV CE certificate (List A) HIV-1 […]
The World Health Organization (WHO) said on May 20 that recently, the organization received reports of about 80 cases of monkeypox from various countries, and another 50 cases are yet to be confirmed. WHO convened an expert and technical advisory group meeting on May 20 to discuss global information on monkeypox and its response. At present, several countries including the […]